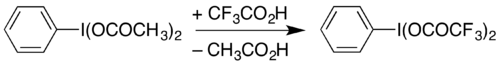(Bis(trifluoroacetoxy)iodo)benzene
(Bis(trifluoroacetoxy)iodo)benzene, Template:Chem, is a hypervalent iodine compound used as a reagent in organic chemistry. It can be used to carry out the Hofmann rearrangement under acidic conditions.[1]
Preparation
The syntheses of all aryl hypervalent iodine compounds start from iodobenzene. The compound can be prepared by reaction of iodobenzene with a mixture of trifluoroperacetic acid and trifluoroacetic acid in a method analogous to the synthesis of Template:Nowrap
It can also be prepared by dissolving diacetoxyiodobenzene (a commercially available compound) with heating in trifluoroacetic acid:[2]
Uses
It also brings around the conversion of a hydrazone to a diazo compound, for example in the diazo-thioketone coupling. It also converts thioacetals to their parent carbonyl compounds.
Hofmann rearrangement
The Hofmann rearrangement is a decarbonylation reaction whereby an amide is converted to an amine by way of an isocyanate intermediate. It is usually carried out under strongly basic conditions.[3][4]
The reaction can also be carried out under mildly acidic conditions by way of the same intermediate using a hypervalent iodine compound in aqueous solution.[1] An example published in Organic Syntheses is the conversion of cyclobutanecarboxamide, easily synthesized from cyclobutylcarboxylic acid, to cyclobutylamine.[2] The primary amine is initially present as its trifluoroacetate salt, which can be converted to the hydrochloride salt to facilitate product purification.[1][2]